Agbr Solid Liquid Or Gas
| |||
| Names | |||
|---|---|---|---|
| IUPAC proper noun Silver(I) bromide | |||
| Other names Bromargyrite | |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.160 | ||
| EC Number |
| ||
| PubChem CID |
| ||
| UNII |
| ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemical formula | AgBr | ||
| Molar mass | 187.77 chiliad/mol | ||
| Appearance | Stake yellow solid photosensitive | ||
| Density | half dozen.473 g/cmthree, solid | ||
| Melting point | 432 °C (810 °F; 705 Thou) | ||
| Boiling point | ane,502 °C (2,736 °F; 1,775 K) (decomposes) | ||
| Solubility in water | 0.140 mg/Fifty (20 °C) | ||
| Solubility product (Thousand sp) | 5.4 × 10 −13 | ||
| Solubility | insoluble in booze, most acids sparingly soluble in ammonia soluble in brine cyanide solutions | ||
| Ring gap | 2.5 eV | ||
| Electron mobility | 4000 cm2/(5·s) | ||
| Magnetic susceptibility (χ) | −59.7·x−vi cm3/mol | ||
| Refractive alphabetize (n D) | 2.253 | ||
| Thermochemistry | |||
| Heat capacity (C) | 270 J/(kg·G) | ||
| Std molar | 107 J·mol−1·K−1 [one] | ||
| Std enthalpy of | −100 kJ·mol−i [1] | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |  | ||
| Signal give-and-take | Alert | ||
| Hazard statements | H410 | ||
| Precautionary statements | P273, P391, P501 | ||
| Related compounds | |||
| Other anions | Silver(I) fluoride Silver chloride Silver iodide | ||
| Other cations | Copper(I) bromide Mercury(I) bromide | ||
| Except where otherwise noted, information are given for materials in their standard land (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Silver bromide (AgBr) is a soft, stake-yellow, h2o-insoluble salt well known (along with other silver halides) for its unusual sensitivity to light. This belongings has immune silver halides to become the basis of mod photographic materials.[2] AgBr is widely used in photographic films and is believed by some to have been used for making the Shroud of Turin.[3] The salt can be establish naturally as the mineral bromargyrite.
Grooming [edit]
Although the compound can be found in mineral form, AgBr is typically prepared by the reaction of silver nitrate with an alkali bromide, typically potassium bromide:[2]
- AgNOiii(aq) + KBr(aq) → AgBr(s)+ KNOiii(aq)
Although less user-friendly, the chemical compound can also be prepared directly from its elements.
Modern preparation of a simple, light-sensitive surface involves forming an emulsion of silver halide crystals in a gelatine, which is so coated onto a film or other back up. The crystals are formed by atmospheric precipitation in a controlled environment to produce minor, uniform crystals (typically < one μm in bore and containing ~1012 Ag atoms) called grains.[2]
Reactions [edit]
Silver bromide reacts readily with liquid ammonia to generate a diversity of ammine complexes, similar Ag(NH
3 )
2 Br and Ag(NH
3 )
two Br −
2 . In general:[4]
- AgBr + m NH3 + (northward - ane) Br −
→ Ag(NH
3 )
m Br one-n
n
Silver bromide reacts with triphenylphosphine to give a tris(triphenylphosphine) production:[5]
Physical properties [edit]
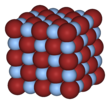
Crystal structure [edit]
AgF, AgCl, and AgBr all have face-centered cubic (fcc) rock-table salt (NaCl) lattice structure with the following lattice parameters:[6]
| Chemical compound | Crystal | Structure | Lattice, a /Å | ||||
| AgF | fcc | rock-common salt, NaCl | 4.936 | ||||
| AgCl, Chlorargyrite | fcc | stone-salt, NaCl | 5.5491 | ||||
| AgBr, Bromargyrite | fcc | stone-salt, NaCl | 5.7745 | ||||
| |||||||
The larger halide ions are arranged in a cubic close-packing, while the smaller silver ions fill up the octahedral gaps between them, giving a six-coordinate construction where a silver ion Ag+ is surrounded by 6 Br− ions, and vice versa. The coordination geometry for AgBr in the NaCl structure is unexpected for Ag(I) which typically forms linear, trigonal (three-coordinated Ag) or tetrahedral (4-coordinated Ag) complexes.
Dissimilar the other silver halides, iodargyrite (AgI) contains a hexagonal zincite lattice structure.
Solubility [edit]
The silver halides have a wide range of solubilities. The solubility of AgF is about half-dozen × ten7 times that of AgI. These differences are attributed to the relative solvation enthalpies of the halide ions; the enthalpy of solvation of fluoride is anomalously large.[7]
| Compound | Solubility (one thousand / 100 g H2O) |
| AgF | 172 |
| AgCl | 0.00019 |
| AgBr | 0.000014 |
| AgI | 0.000003 |
Photosensitivity [edit]
Although photographic processes had been in use since the mid-1800s, there were no suitable theoretical explanations until 1938 with the publication of a paper by R.W. Gurney and N.F. Mott.[8] This paper triggered a large amount of research in fields of solid-state chemistry and physics, also more than specifically in silver halide photosensitivity phenomena.[2]
Further research into this mechanism revealed that the photographic backdrop of silver halides (in item AgBr) were a consequence of deviations from an ideal crystal construction. Factors such as crystal growth, impurities, and surface defects all bear upon concentrations of indicate ionic defects and electronic traps, which bear on the sensitivity to calorie-free and allow for the formation of a latent image.[iii]
- Frenkel defects and quadropolar deformation
The major defect in silver halides is the Frenkel defect, where silver ions are located interstitially (Agi +) in high concentration with their corresponding negatively charged silver-ion vacancies (Agv −). What is unique nigh AgBr Frenkel pairs is that the interstitial Agi + are uncommonly mobile, and that its concentration in the layer below the grain surface (called the space-charge layer) far exceeds that of the intrinsic bulk.[3] [9] The formation energy of the Frenkel pair is low at 1.xvi eV, and the migration activation energy is unusually low at 0.05 eV (compare to NaCl: 2.eighteen eV for the formation of a Schottky pair and 0.75 eV for cationic migration). These low energies issue in big defect concentrations, which tin reach near 1% almost the melting point.[ix]
The low activation energy in silver bromide can be attributed the silver ions' high quadrupolar polarizability; that is, information technology tin hands deform from a sphere into an ellipsoid. This property, a effect of the d9 electronic configuration of the silvery ion, facilitates migration in both the silver ion and in silverish-ion vacancies, thus giving the unusually depression migration free energy (for Agv −: 0.29–0.33 eV, compared to 0.65 eV for NaCl).[nine]
Studies take demonstrated that the defect concentrations are strongly affected (up to several powers of 10) past crystal size. Most defects, such as interstitial silver ion concentration and surface kinks, are inversely proportional to crystal size, although vacancy defects are straight proportional. This phenomenon is attributed to changes in the surface chemistry equilibrium, and thus affects each defect concentration differently.[3]
Impurity concentrations can be controlled by crystal growth or direct add-on of impurities to the crystal solutions. Although impurities in the silverish bromide lattice are necessary to encourage Frenkel defect formation, studies by Hamilton have shown that above a specific concentration of impurities, the numbers of defects of interstitial silver ions and positive kinks reduce sharply by several orders of magnitude. Afterward this point, only silvery-ion vacancy defects, which actually increment by several orders of magnitude, are prominent.[3]
- Electron traps and hole traps
When low-cal is incident on the silver halide grain surface, a photoelectron is generated when a halide loses its electron to the conduction band:[2] [3] [x]
- 10− + hν → Ten + e−
Afterwards the electron is released, information technology will combine with an interstitial Agi + to create a silver metallic atom Agi 0:[2] [3] [ten]
- east− + Agi + → Agi 0
Through the defects in the crystal, the electron is able to reduce its energy and become trapped in the atom.[2] The extent of grain boundaries and defects in the crystal bear on the lifetime of the photoelectron, where crystals with a big concentration of defects will trap an electron much faster than a purer crystal.[10]
When a photoelectron is mobilized, a photohole h• is as well formed, which also needs to be neutralized. The lifetime of a photohole, however, does not correlate with that of a photoelectron. This detail suggests a dissimilar trapping mechanism; Malinowski suggests that the pigsty traps may be related to defects as a effect of impurities.[10] One time trapped, the holes attract mobile, negatively charged defects in the lattice: the interstitial silvery vacancy Agv −:[10]
- h• + Agfive − ⇌ h.Agv
The formation of the h.Agv lowers its free energy sufficiently to stabilize the circuitous and reduce the probability of ejection of the hole back into the valence ring (the equilibrium constant for hole-complex in the interior of the crystal is estimated at 10−4.[10]
Additional investigations on electron- and hole-trapping demonstrated that impurities too can be a significant trapping system. Consequently, interstitial silverish ions may not be reduced. Therefore, these traps are actually loss mechanisms, and are considered trapping inefficiencies. For instance, atmospheric oxygen can collaborate with photoelectrons to form an O2 − species, which can collaborate with a pigsty to reverse the complex and undergo recombination. Metal ion impurities such as copper(I), fe(II), and cadmium(II) take demonstrated hole-trapping in silver bromide.[3]
- Crystal surface chemistry;
Once the hole-complexes are formed, they lengthened to the surface of the grain as a issue of the formed concentration gradient. Studies demonstrated that the lifetimes of holes almost the surface of the grain are much longer than those in the majority, and that these holes are in equilibrium with adsorbed bromine. The internet effect is an equilibrium push at the surface to form more holes. Therefore, every bit the hole-complexes reach the surface, they disassociate:[x]
- h.Ag5 − → h• + Agv − → Br → FRACTION Br2
By this reaction equilibrium, the pigsty-complexes are constantly consumed at the surface, which acts as a sink, until removed from the crystal. This mechanism provides the analogue to the reduction of the interstitial Agi + to Agi 0, giving an overall equation of:[10]
- AgBr → Ag + FRACTION Br2
- Latent epitome germination and photography
To summarize, as a photographic film is subjected to an image, photons incident on the grain produce electrons which interact to yield silver metallic. More photons hitting a detail grain volition produce a larger concentration of silver atoms, containing between 5 and 50 silver atoms (out of ~1012 atoms), depending on the sensitivity of the emulsion. The moving-picture show now has a concentration gradient of silvery atom specks based upon varying intensity light beyond its area, producing an invisible "latent image".[2] [ten]
While this process is occurring, bromine atoms are existence produced at the surface of the crystal. To collect the bromine, a layer on top of the emulsion, chosen a sensitizer, acts as a bromine acceptor.[10]
During movie evolution the latent image is intensified by add-on of a chemical, typically hydroquinone, that selectively reduces those grains which contain atoms of silver. The process, which is sensitive to temperature and concentration, will completely reduce grains to silvery metal, intensifying the latent paradigm on the club of 1010 to 10xi. This footstep demonstrates the advantage and superiority of silver halides over other systems: the latent epitome, which takes merely milliseconds to grade and is invisible, is sufficient to produce a total image from it.[two]
Later on development, the film is "fixed," during which the remaining silvery salts are removed to prevent further reduction, leaving the "negative" image on the film. The agent used is sodium thiosulfate, and reacts according to the following equation:[2]
- AgX(southward) + 2 Na2Southward2O3(aq) → Na3[Ag(SouthwardtwoOiii)2](aq) + NaX(aq)
An indefinite number of positive prints can be generated from the negative by passing low-cal through it and undertaking the same steps outlined above.[2]
Semiconductor backdrop [edit]
As silver bromide is heated within 100 °C of its melting point, an Arrhenius plot of the ionic conductivity shows the value increasing and "upward-turning." Other physical properties such as elastic moduli, specific heat, and the electronic energy gap also increase, suggesting the crystal is budgeted instability.[9] This behavior, typical of a semi-conductor, is attributed to a temperature-dependence of Frenkel defect formation, and, when normalized confronting the concentration of Frenkel defects, the Arrhenius plot linearizes.[9]
Run across too [edit]
- Photography
- Science of photography
- Silver chloride
References [edit]
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Visitor. p. A23. ISBN978-0-618-94690-7.
- ^ a b c d due east f k h i j g Greenwood, N.N., Earnshaw, A. (1984). Chemistry of the Elements. New York: Permagon Press. pp. 1185–87. ISBN978-0-08-022057-4.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b c d due east f chiliad h Hamilton, J.F. (1974). "Physical Backdrop of Silver Halide Microcrystals". Photographic Scientific discipline and Engineering science. 18 (5): 493–500.
- ^
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Engelhardt, LM; Healy, PC; Patrick, VA; White, AH (1987). "Lewis-Base Adducts of Group-11 Metal(I) Compounds. Xxx. 3:one Complexes of Triphenylphosphine With Argent(I) Halides". Aust. J. Chem. forty (11): 1873–1880. doi:x.1071/CH9871873.
- ^ Glaus, Due south. & Calzaferri, G. (2003). "The band structures of the argent halides AgF, AgCl, and AgBr: A comparative study". Photochem. Photobiol. Sci. 2 (iv): 398–401. doi:10.1039/b211678b.
- ^ Lide, David R. (ed). (2005)Handbook of Chemistry and Physics, 86th Edition, The Chemic Condom Publishing Co., Cleveland.
- ^ Gurney, R. W.; Mott, N. F. (1938). "The theory of the photolysis of silver bromide and the photographic latent paradigm". Proc. Roy. Soc. A164 (917): 151–167. Bibcode:1938RSPSA.164..151G. doi:x.1098/rspa.1938.0011.
- ^ a b c d due east Slifkin, L. M. (1989). "The Physics of Lattice Defects in Argent Halides". Crystal Lattice Defects and Amorphous Materials. xviii: 81–96.
- ^ a b c d east f g h i j Malinowski, J. (1968). "The Role of Holes in the Photographic Process". The Journal of Photographic Science. 16 (2): 57–62. doi:10.1080/00223638.1968.11737436.
Agbr Solid Liquid Or Gas,
Source: https://en.wikipedia.org/wiki/Silver_bromide
Posted by: osorioappithe.blogspot.com





0 Response to "Agbr Solid Liquid Or Gas"
Post a Comment